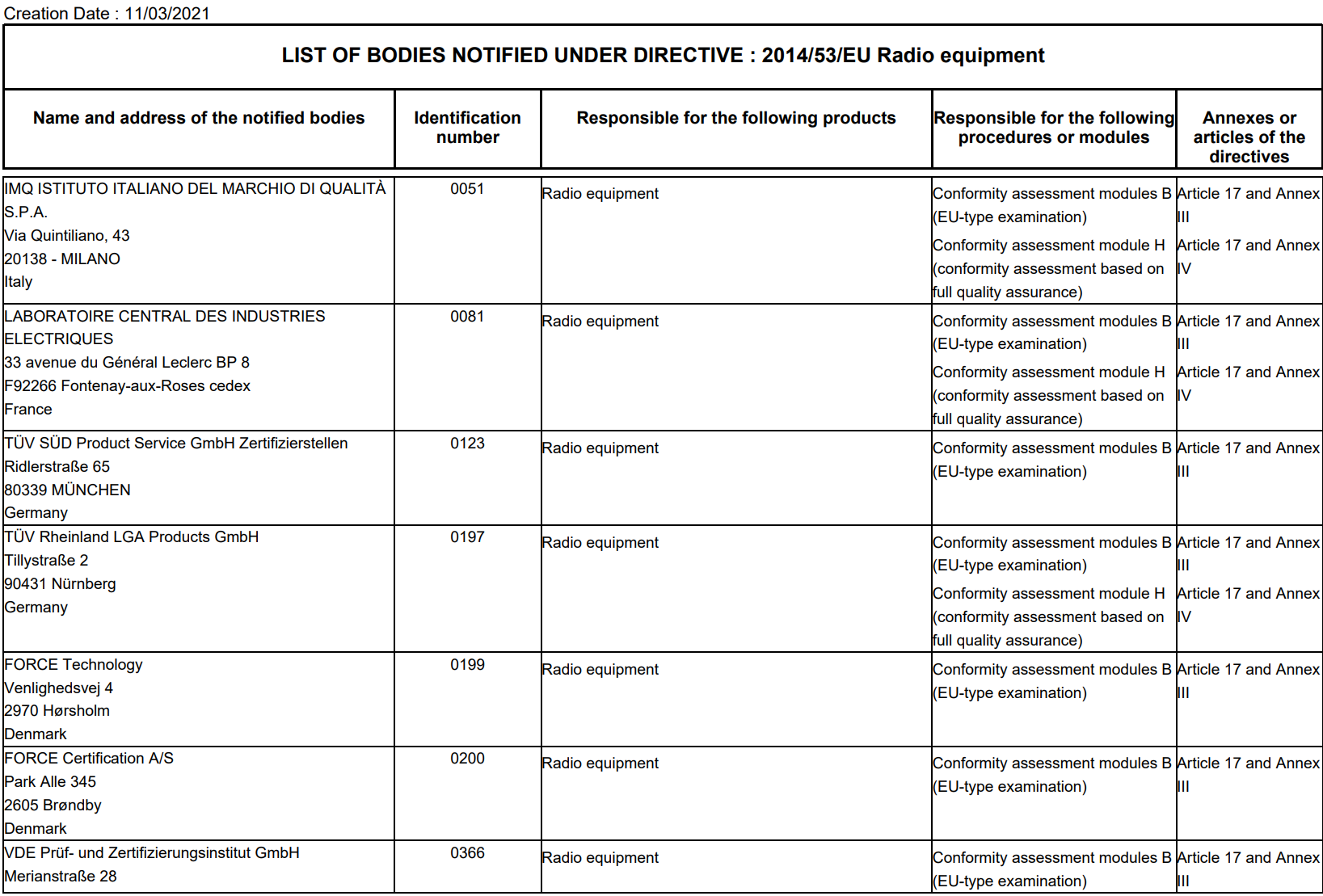
Which EU Notified Bodies Have Been “Designated” Under the MDR 2017/745 and IVDR 2017/746? – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog

Mario Gabrielli Cossellu on LinkedIn: #notifiedbody #mdr #nando #ivdr #conformityassessment #eu #regulations… | 11 comments
Availability and capacity of notified bodies to carry out conformity assessments for COVID-19 related medical devices and in vit
Availability and capacity of notified bodies to carry out conformity assessments for COVID-19 related medical devices and in vit